 
For advice on revaccination frequency and annual booster vaccinations, consultation with a veterinarian is recommended. The need for this booster has not been established. Historically, annual revaccination with a single dose has been recommended for this product. The presence of maternal antibody is known to interfere with the development of active immunity in dogs and additional boosters will be required in most young animals. All dogs should initially receive one dose of this product and a second dose 2 to 3 weeks later. If blood enters the syringe freely, choose another injection site. IMPORTANT NOTE: Before injecting vaccine, pull back slightly on syringe plunger. Do not inject directly into blood vessel (see note below). Subcutaneous administration: Lift the loose skin behind the neck or behind the front leg and insert needle (see illustration, arrows 1 and 2). Withdraw entire contents into the syringe. Gentamicin added as preservative.ĭirections and dosage: One Dose - Open the syringe by twisting or tapping the cap against a hard surface to break the heat weld.Īseptically rehydrate the vaccine cake with 1 mL of sterile diluent supplied. This product contains a CPV 2b strain which has been demonstrated effective against disease caused by CPV 2c in puppies with maternal antibody. CompositionĬanine Spectra ® 5 vaccine is a combination of immunogenic, attenuated strains of CDV, CAV2, CPIV, and CPV type 2b, propagated in cell line tissue cultures The CAV2 fraction has been shown to be effective against disease due to CAV1. For more information regarding efficacy and safety data go to. The duration of immunity of this product has not been determined. This product has been shown to be effective for the vaccination of healthy dogs 6 weeks of age or older against canine distemper virus (CDV), infectious canine hepatitis (CAV1), canine adenovirus type 2 (CAV2), canine parainfluenza (CPIV), and canine parvovirus (CPV). KEEP REFRIGERATED Canine Spectra 5 Indications.Produced by: Elanco US Inc., Fort Dodge, IA 50501 USAĭistributed by: Peak Marketing, Blue Springs, MO 64014 USAĬanine Spectra ® is a registered trademark of Durvet Inc.Canine Distemper-Adenovirus Type 2-Parainfluenza-Parvovirus Vaccine Inactivate unused contents before disposal. In case of human exposure, contact a physician. This product has not been tested in pregnant animals. In case of anaphylactoid reaction, administer epinephrine. While these are usually temporary signs, consultation with a veterinarian may be indicated if they persist or progressively worsen. A very small percentage of dogs may show sneezing, coughing, or nasal discharge following vaccination. 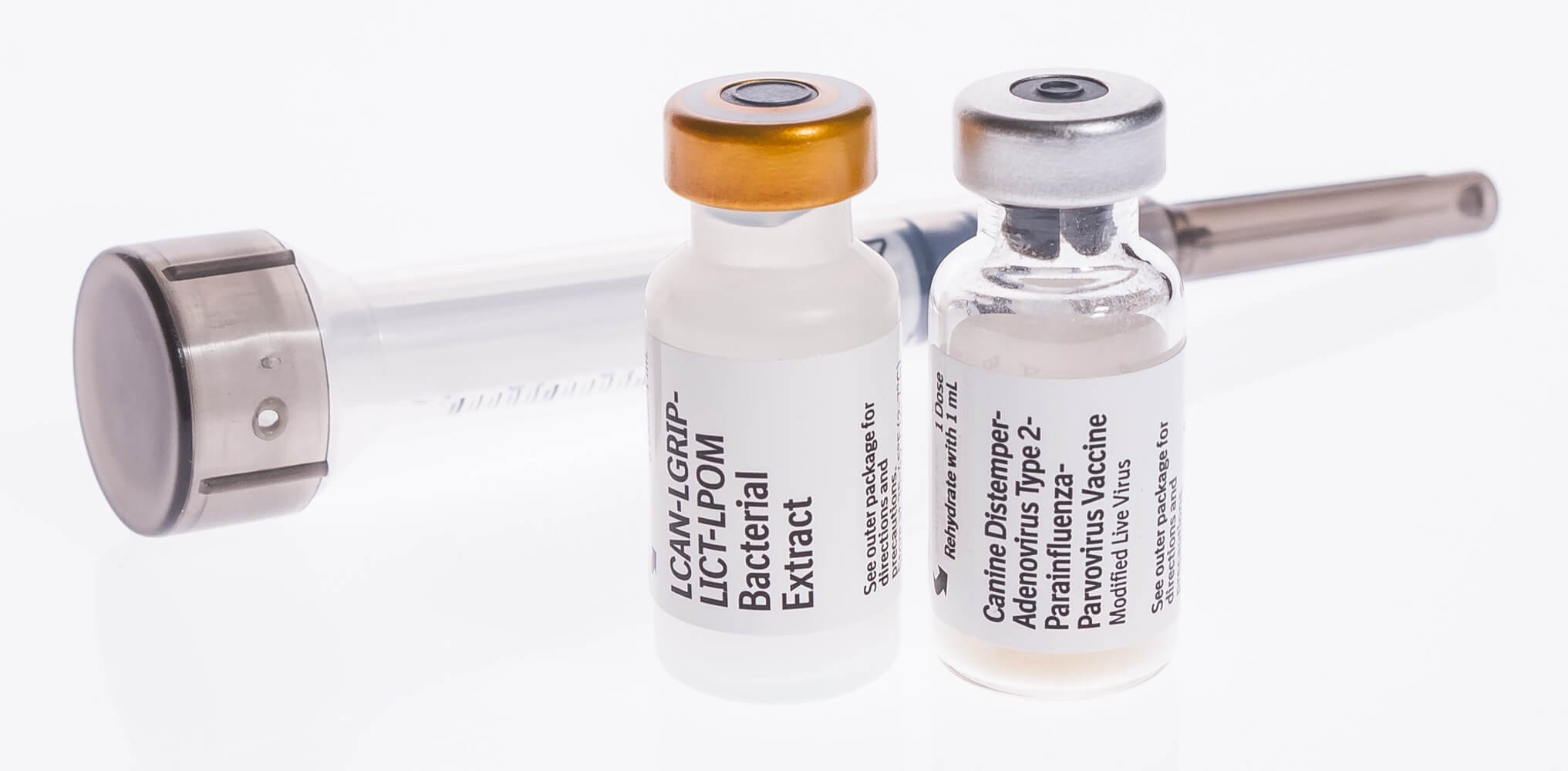
Do not mix with other products, except as specified on the label. Precautions FOR INTRANASAL USE IN DOGS ONLY. Vaccines that protect against Enteric Canine Coronavirus infection do not provide protection against the respiratory form of this disease. Historically, annual revaccination with this product has been recommended. Disease Overview There are two forms of Canine Coronavirus: Enteric Canine Coronavirus (CCoV) and Respiratory Canine Coronavirus (CRCoV). Using the syringe with the applicator tip attached, administer 0.5 mL of rehydrated vaccine into each nostril. Twist and remove needle from syringe and attach enclosed applicator tip. 
Aseptically rehydrate the vaccine cake with 1 ml of sterile diluent supplied. DescriptionĬanine Spectra KC3™ vaccine is a combination of immunogenic, attenuated strain of CAV2 and CPIV propagated in cell line tissue cultures with an avirulent live culture of B.bronch.ĭirections and dosage: Open the syringe by twisting or tapping the cap against a hard surface to break the heat weld. The duration of immunity has not been determined. This product has been shown to be effective for the vaccination of healthy dogs 8 weeks of age or older against Bordetella bronchiseptica (B.bronch), canine parainfluenza (CPIV), and canine adenovirus type 2 (CAV2). Protects against all known strains of canine parvovirus (CPV), including CPV-2c 3,4. KEEP PRODUCT REFRIGERATED Canine Spectra KC3 Indications The only 0.5 mL combination vaccine formula to include parainfluenza. Canine Adenovirus Type 2-Parainfluenza-Bordetella Bronchiseptica Vaccine 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
